Studies document substantial differences of GM maize and GM soybean from their conventional non-GM counterparts, exposing a permissive regulatory regime that has failed miserably in protecting public health and biodiversity Dr Eva Sirinathsinghji
Several new studies carried out by scientists independent of the biotech industry are showing up glaring differences between GMOs and their non-GMO counterparts. This makes a mockery of the regulatory principle of ‘Substantial Equivalence’ which has facilitated approvals of GMOs with practically no protection for public health and the environment [1] (see [2] The Principle of Substantial equivalence is Unscientific and Arbitrary, ISIS news).
The concept of ‘Substantial Equivalence’ was first introduced in 1993 by the Organisation for Economic Development (OECD), an international economic and trade organisation, not a public health body. The principle states that if a new food is found to be substantially equivalent to an already existing food product, it can be treated the same way as the existing product with respect to safety. This concept has greatly benefited the trade of GM produce, allowing it to effectively bypass regulatory requirements that would apply to novel food and other products including novel chemical compounds, pharmaceuticals, pesticides and food additives, all of which require a range of toxicological tests and can be subject to legal limitations on safe consumption/intake.
Regulatory agencies including the US Food and Drug Administration, the Canadian Food Inspection Agency and Japan’s Ministry of Health and Welfare, generally base their GM food safety regulations on substantial equivalence.
There are many good reasons for consumers to feel unprotected by these regulatory policies, not least because the principle itself is designed to be as flexible and open to interpretation for the approval of just about any and every GMO submitted. In practice, the principle allows the comparison of a GM line to any existing variety within the same species, and even to an abstract entity made up of ingredients from a collection of species. This means that a GM variety can have all the worst traits of many different varieties and still be deemed substantially equivalent [1, 2]. Traits used for comparisons are also based solely on gross and insensitive chemical compositional tests such as levels of carbohydrate, protein and sugars. This process cannot even begin to tackle safety issues. Ironically, for the GMOs to be patentable as they are, a clear novelty, i.e., a difference or non-substantial equivalence is indeed required.
Independent assessments of substantial equivalence have shown how this ill-defined practice is not only inadequate but untrustworthy [3- 5], and the new studies most clearly confirm this.
In April 2013, an Egyptian publication led by Professor El-Sayed Shaltout at Alexandria University found that Monsanto’s 810 Corn (Ajeeb-YG®), modified to express the insecticidal Bt Cry1Ab gene, has increased total protein, crude fat, crude fibre & total saccharides and decreased starch content compared with non-GM Ajeeb corn. Abnormal levels of certain amino acids, fatty acids and elements were also recorded [6]. These compositional differences only gave the merest hint of the toxicity of the GM corn revealed in previous male rat feeding studies conducted by the same team documenting a wide range of organ and tissue abnormalities [7, 8]. Liver cells displayed vacuolation and fatty degeneration. The kidneys had congested blood vessels and dilation of renal tubules. The testes showed signs of necrosis and desquamation of spermatogoneal germ cells lining the seminiferous tubules. The spleens were congested with slight lymphocytic depletion. The small intestines showed hyperplasia and hyperactivation of mucous secretory glands, with necrosis of intestinal villi. Most certainly, the GM corn was not substantially equivalent to non-GM corn.
A more recent study led by Thomas Bøhn at the Norwegian Centre for Biosafety [9] tested 31 batches of whole soybeans from Iowa, US in three categories: 1) GM glyphosate-tolerant soy; 2) unmodified soy cultivated using conventional “chemical” regime and 3) unmodified organically cultivated soy. The three groups were analysed for chemical contamination (organochlorine, organophosphorus, pyrethroides, PCBs, glyphosate and AMPA (aminomethylphosponic acid - the major degradation product of glyphosate) based on the list of pesticide brand names used by the farmers) as well as nutritional content. Testing pesticide levels is important as substantial equivalence assessments for GM glyphosate-tolerant soy were not previously done with herbicide residue in the crop despite common knowledge that glyphosate is actually taken into the plant, and also alters the metabolism and biochemistry, and hence the chemical composition of crops. Any assessment of its equivalence is obviously irrelevant when glyphosate is not included.
The results couldn’t be clearer. As shown in Figure 1, glyphosate and AMPA were only present in GM soy samples and not at all in conventional non-GM and organic varieties. In the GM-soy samples, the concentration of AMPA (mean concentration = 5.74 mg/kg) was on average nearly twice as high as glyphosate (3.26 mg/kg). Other herbicides were detected: Fluazifop-P a selective phenoxy herbicide, was found at a concentration of 0.078 mg/kg in one of the GM-soy samples, malathion was found at a concentration of 0.02 mg/kg in one of the conventional soy samples and Dieldrin was found at a concentration of 0.002 mg/kg in one of the organic soy samples. No other residues were detected. Additional testing for pesticide residues in pooled samples of GM, conventional and organic soybeans showed trace-levels of Alpha-endosulfane, Trans-nonachlor and Trans-chlordane, all close to the detection limit of 0.05 µg/kg and in all soy types. Dieldrin was also found in very low levels with 0.51, 0.45 and 0.6 µg/kg in GM, conventional and organic soybeans, respectively.
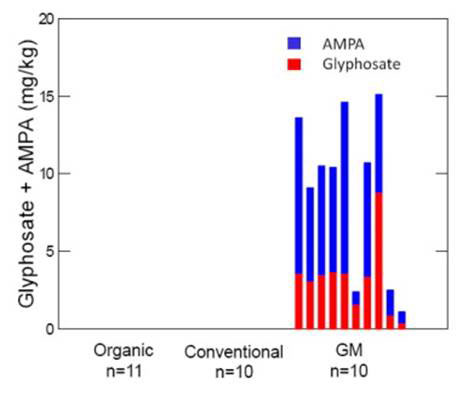
Figure 1 Gyphosate and its metabolite AMPA present only in GM soy
The researchers looked at chemical composition of the soy samples including composition of protein, fat and sugar content, as well as individual amino acids, vitamins, fatty acids and elements. Organic soy samples show significant differences from both GM and conventional non-GM soybean samples, with higher and lower levels of protein and saturated fats respectively, plus significant differences in levels of total as well as individual levels of amino acids, vitamins, and minerals. A further statistical multivariate analysis of the compositional results found without exception that each individual soybean sample could be discriminated statistically into their respective agricultural background, even excluding the data on glyphosate/AMPA levels. The organic soybean was nutritionally superior to both conventionally grown non-GM soybean and GM soybean.
Profiling technologies, such as proteomics, allow the simultaneous measurement and comparison of thousands of plant components, in this case proteins, without prior knowledge of their identity. These methods are now being employed by independent scientists to provide a more thorough, unbiased and global profile of GM crop composition for risk assessment.
A new study conducted in Brazil by Agapito-Tenfen and colleagues at the Federal University of Santa Catarina is an example of this type of analysis, with global protein expression analysed in GM MON810 compared with the non-GM maize control grown in two different environmental conditions. Analysis of the total leaf-derived proteome showed 32 differentially expressed proteins (out of an average 458 and 643 detected proteins for each condition) between GM and non-GM maize with most of them involved in carbohydrate metabolism, stress response as well as genetic information processing such as post-translational modification of newly made proteins [10]. Sixteen proteins were differentially expressed between GM and non-GM maize at each of the two growing locations (Campos Novos and Chapecó). In Campos Novos, the experiment found 8 proteins detectable only in the GM samples, the remaining 8 were absent in the GM samples. In Chapecó, there were seven proteins exclusive to GM plants and seven to non-GM plants. 2 proteins showed quantitative differences in expression. For example, glyceraldehydes 3-phosphate degydrogenase (GAPDH) and fructose-biphosphate, ferredoxin-NAPD was exclusive to GM plants in Capos Novos and relate directly to energy metabolism. When it comes to carbohydrate metabolism, this is consistent with previous studies that found increased sugar levels in MON810 plants, with up 14, 7 and 1.8-fold increases in glucose, fructose and sucrose respectively [11]. Indeed, maize plants go through many developmental stages in their leaves that exclusively rely on carbohydrate metabolism. Further, transgenes with high constitutive promoters have been shown to have a high energetic cost e.g. cauliflower mosaic virus 35S promoter [12, 13], which the authors speculate may cause a problem for transgenic plants.
Stress response genes e.g. those related to glutathione metabolism (glyoxylase 1 and IN2-1), peroxidises and pathogenesis-related protein were expressed only in non-GM plants. It was also revealed that 2-cysteine peroxiredoxin BAS1 (2-CP) proteins are over-expressed in GM plants from both locations. Peroxidases are of great importance for eliminating H2O2 resulting from oxidative phosphorylation.
Four genetic information processing proteins were differentially expressed. Two of these were only present in GM plants from Campos Novos, the adenine phosphoribosyl transferase (APT), and the ATP-dependent Clp protease ATP-binding subunit ClpA (Clp-ClpA). APT works on adenine salvage in plants, while Clp-ClpA proteases exert unfoldase activity, playing a key role in regulating the availability of certain short-lived regulatory proteins. Chaperonin protein and S-adenosylmethionine synthetase 1 were upregulated in non-GM plants. S-adenosylmethionine synthetase 1 is involved in transmethylation of proteins, nucleic acids, polysaccharides and fatty acids. Interestingly, many of these genetic information processing proteins are directly related to gene expression control.
This study is the first of its kind to use such technologies to assess how both the environment and genotype can influence plant composition in Brazil and highlights the routine profiling analyses now widely available for proteins, transcripts and metabolites that are still not required by governments for regulatory approval as they should be.
Of further note in this study is the effect of environmental conditions on the composition of crops. GM proponents often argue that other factors such as environmental conditions as well as hybrid varieties determine the composition and physiology of a plant but genetic modification can influence such conditions. Indeed, the environment did cause variation in composition of the crops, but interestingly, it appeared that the GM maize protein expression profile was more affected by the environment.
To conclude, the numerous differences demonstrated between GM varieties and their non-GM counterpart may well impact consumer health and biodiversity, and clearly exposes the substantial equivalence principle as pseudoscience. In reality, genetic modification causes very real and substantial, unpredictable and uncontrollable changes in the host genome including mutations, and rearrangements as well as new transcripts and proteins. Further, glyphosate and GM crops have already been shown to cause damage to both health and the environment in many independent studies (see [14] Ban GMOs Now, ISIS special report). This is now fully confirmed in the new studies.
Article first published 19/02/14
Comments are now closed for this article
There are 5 comments on this article.
Pietro Perrino Comment left 20th February 2014 06:06:20
Dear Mae Wan, I am reading "Frankenstein's Cat - Cuddling Up to Biotech's Brave New Beasts", 2013, by Emily Anthes. The book tries to convince the reader that genetic engineering is the right tool for the future. I am sure you have head of it. I would like to know your thought.
Rory Short Comment left 20th February 2014 22:10:16
The 'substantial equivalence principle' is from a scientific point of view completely meaningless. Equivalence of what to what? It does not say. And what are the measurements that are being compared? It cannot say because it does not say what is to be measured and how it is to be measured. It is a phrase dreamed up by marketers not scientists and the word 'principle' was particularly included in it in order to give, fraudulently, a veneer of science to the phrase.
Theresa Comment left 21st February 2014 21:09:14
GM foods and application of the precautionary principle in Europe
Link to consultation, deadline noon 23 April:
http://www.parliament.uk/business/committees/committees-a-z/commons-select/science-and-technology-committee/inquiries/parliament-2010/gm-foods-and-application-of-the-precautionary-principle-in-europe/commons-written-submission-form/
Theresa Comment left 21st February 2014 21:09:00
Following on from Pietro's comment, I have just listened to Emily Anthes being interviewed. She appears to contradict herself, admits there are important areas that she does not know about... What are my thoughts? It would be impolite to say.
Listening to her was excruciatingly painful.
Luckily, as you will see from the comments on this link, others (majority) are not so naive as her.
http://www.wnyc.org/story/276125-franken-critters/
Todd Millions Comment left 25th February 2014 19:07:29
interesting article further to this;by Antony Samsel and Stephanie Seneff- Glyphosate pathways to modern diseases II:Celiac sprue and gluten intolerance from;Interdisilpinary toxicology 2013 vol6.The graphs are paticularly-
Florence Nightengale grade- Striking.
Best enjoyed on an empty stomach.If search doesn't bring it up, go to Globalresearch.ca
Cheers-Hope land is now in view.