I-SIS Special Miniseries
Save Our Oceans, Save Our Planet
Dr. Mae-Wan Ho explains how oceans determine climate and influence climate change. Urgent need to shift away from fossil fuels to renewable options (Which Energy?)
Oceans are vast, deep, and mysterious. They cover 71 percent of the earth’s surface and contain approximately 97 percent of the earth’s water [1]. The average depth of the ocean is almost 4 000 metres [2], and nearly half the area is over 3 000 metres deep.
The oceans are home to the majority of plant and animal life on earth, accounting for 90 percent of the world’s living biomass, and many new species are being discovered from the depths. The Census of Marine Life (COML), an international alliance of scientists from 70 countries, discovered some 13 000 new species in 2003 alone [3]. COML is amassing data to create a map of the distribution of 38 000 marine species from plankton to whales.
According to one theory, life on earth originated around hydrothermal vents in the deep seafloor that heat up the water to 450 C [2]. Rare creatures living there today are still able to obtain energy from chemical reactions in the same way as the primordial life forms, and do not need to depend directly or indirectly on photosynthesis for food like 99.97 percent of the biosphere.
Oceans affect climate in many ways [4]. As the major reservoir of water, oceans dominate the movement of water, supplying most of the water vapour in the atmosphere by evaporation. Of this evaporated water, 91 percent is returned to the oceans as precipitation, the remainder is transported and precipitated over landmasses. Runoff and groundwater from land flow back to the oceans [5].
The oceans and the atmosphere are tightly linked, and together form the most dynamic component of the earth’s climate system. They bring moisture to coastal areas that may be carried inland by the wind. Typhoons and hurricanes form over the oceans, and as oceans get warmer these will be more frequent.
Oceans store heat. When the earth’s surface cools or is heated up by the sun, the temperature change is greater and faster over land than over the oceans. One consequence of the ocean’s ability to absorb more heat is that it cools the surrounding when it is hot and warms it when it is cold, which is why maritime climates tend to be less extreme than continental.
Winds and currents are constantly moving the ocean’s waters. Surface currents flowing north or south transport heat, and carry warmed or cooled water several thousand kilometres, thereby ameliorating the extremes of heat or cold. Deep ocean currents also flow across the globe [6] (Why Gaia needs rainforests, SiS 20). The Gulf Stream Drift, for example, is powered by cold, dense, salt-laden water sinking off the north polar coastal regions and moving south in the depths, pushing the surface warm water from the tropical and subtropical Atlantic (including some from the Gulf of Mexico) up north to bathe the shores of Western Europe, producing a climate that is surprisingly mild for that latitude.
Global warming and melting of the polar icecaps freshens the surface water, reducing its density and preventing it from sinking (Global Warming is happening, this issue). As a result, the Gulf Stream slows down, or may even reverse, bringing severe winters to northern Europe while the rest of the earth heats up [7] (Global warming and then the big freeze, SiS 20). In places where cold deep waters come up to the surface, as near San Francisco in California USA, the climate is as cool as Dublin in Ireland 1 600 km further north.
Oceans play a fundamental role in climate change through the Carbon Cycle (Box 1). The major driver of the Carbon Cycle is the biosphere, which depends entirely on the ability of green plants, algae and certain bacteria to produce food for themselves and other organisms through photosynthesis. For that reason they are called primary producers.
Box 1
The Carbon Cycle
The carbon cycle supports life on earth and keeps its climate stable. Carbon is the fourth most abundant element on earth, and makes up 50 percent of the dry weight of living organisms [4]. The global carbon cycle involves the flow of carbon between the major carbon reservoirs: the atmosphere, the oceans, the vegetation and soils of terrestrial ecosystems, and fossil fuels deposits (Fig. 1).
As can be seen, huge amounts of carbon are stored in the oceans (especially the deep oceans), in the fossil fuels reserves, and the soils, compared with what’s in the atmosphere. There is no returning arrow to the fossil fuels to balance the outflow, at least not over timescales shorter than millions of years, which means that the carbon released into the atmosphere cannot be reabsorbed. In addition, change in land use also releases the carbon stored in old forests over thousands of years into the atmosphere.
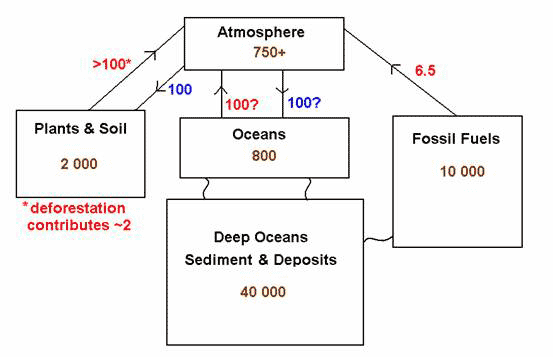
Figure 1. The Carbon Cycle
Standing carbon stocks in Gt C, carbon entering and leaving the atmosphere in Gt C per year
The major driver of the carbon cycle is the biosphere, especially on time scales of hours, days, to thousands of years. Carbon is assimilated into the biosphere through photosynthesis, which turns carbon dioxide from the atmosphere into sugars that provide the building blocks and energy for all life forms. Carbon dioxide is returned to the atmosphere when complex molecules are broken down in respiration to provide energy for living activities.
The amount of carbon taken up by photosynthesis and released back to the atmosphere by respiration each year is 1 000 times greater than the amount of carbon flowing through the geological carbon cycle, which operates over times scales of millions of years. Carbon dioxide dissolved in water forms a weak carbonic acid, which slowly combines with calcium and magnesium in the earth’s crust to form insoluble carbonates, a process called weathering. Then through erosion, the carbonates are washed into the oceans and eventually settle to the bottom. The cycle continues as these materials are folded into the deep layers of the earth and the carbon is the returned to the atmosphere as carbon dioxide during volcanic eruptions.
The biosphere also plays a role in the geological cycle. Land vegetation enhances weathering of soil and the slow uptake of carbon dioxide from the atmosphere to form carbonates. In the oceans, some of the carbon taken up by phytoplankton (microscopic marine plants that form the basis of the marine food web), zooplankton, and other organisms to make calcium carbonate shells settles to the bottom of the ocean to form sediments (see Box 2). Over millions of years when photosynthesis exceeded respiration, organic matter accumulated to form coal and oil deposits, which removed carbon dioxide from the atmosphere to carbon stores in geologic sediments. Burning fossil fuels thereby destroys carbon stores that took millions of years to build up.
There is no doubt that human activities have thrown the carbon cycle out of balance, and carbon dioxide levels have risen 30 percent since the industrial revolution around the middle of the 19th century. Carbon dioxide is the predominant greenhouse gas that raises the temperature of the earth, causing a general disruption of the climate.
Most of the increase in atmospheric carbon dioxide concentrations came from the use of fossil fuels, but about 25 percent came from changes in land use, as for example, through clearing forests for intensive agriculture. Scientists at Woods Hole Research Center showed that between 1859 and 2000, about 155 Gt of C were released to the atmosphere from changes in land use worldwide, the amount released each year generally increased over the period, and by the 1990s, the rate of release averaged about 2 Gt C per year [8].
On land, the primary producers are almost all higher plants: trees, shrubs, grasses, ferns, mosses. In the oceans, however, the primary producers are algae, especially microscopic phytoplankton floating in the surface layers of water [9] (see Box 2).
Although comprising only 0.2 percent of the earth’s total biomass, phytoplankton is responsible for nearly 50 percent of the earth’s primary production on account of its prodigious rate of growth, which is why it can support the incredibly rich and diverse marine community from zooplankton to whales.
Box 2
Phytoplankton
Phytoplankton consists of single-celled algae of three main groups, diatoms, coccolithophores, and dinoflagellates. Diatoms have a pillbox-like outer shell made of silica. As silica is a relatively insoluble in seawater, diatoms form a major component of the sediments in areas of high productivity, most notably in a belt around the Antarctic and in the North Pacific. If the sediment is at least 30 percent diatom shells, it is known as diatomaceous ooze. The silica shells are very ornamental. Centric diatoms are round, and pennates are elongated. Some species form chains. Many have spines, increasing the surface area of the cell and retarding predation. Coccolithophores are coated with calcium carbonate plates or discs called coccoliths. When the cell dies, the coccoliths sink to the bottom and contribute to the sediments. They are usually quite small. Dinoflagellates typically have an outer covering of cellulose plates that fit together like a jigsaw puzzle. Cellulose is organic, and does not contribute to the sediments. Dinoflagellates have flagella, whip-like structures used for motility. Some dinoflagellates are bioluminescent, producing “living light” while others are responsible for ‘red tides’ - toxic algal blooms produced in polluted water that’s over-rich in nutrients.
Phytoplankton inhabits the top 100 m or so of seawater, limited by the availability of light. Photosynthesis can still take place at 1 percent of the light level and phytoplankton grows best not right at the surface, but further down at about 100 m where the water is richer in nutrients such as nitrogen, phosphorous and iron.
Most of the carbon in the upper ocean is recycled, but some of it sinks into the deeper waters to feed the other organisms or to become buried in the seafloor. And as organisms die, their bodies also sink to the seafloor where decomposers feed on them to release carbon dioxide and other nutrients. As a result the deep waters are rich in nutrients.
In certain regions of the ocean, currents interacting with the coast or with other currents or both bring the cold deep nutrient-rich ocean water to the surface. This ‘upwelling’ is important for keeping the phytoplankton productive.
The latest evidence, obtained by towing a video camera submerged in the surface layers of the ocean, suggests that nitrogen-fixing cyanobacteria growing there may be supplying up to 50 percent of the nitrogen required for the phytoplankton [10].
The enormous food web in the oceans that depend on phytoplankton makes phytoplankton the most important primary producer. It is unique among primary producers for growing at the fastest rates, with doubling times from hours to days depending on the conditions [11]. Because of its prodigious rate of growth, phytoplankton population also determines carbon balance in the oceans, which will impact on climate change (Oceans carbon sink or source? this series).
For phytoplankton to thrive there must be an appropriate supply of nutrients. Too little, and the phytoplankton will fail to grow, too much all at once from agricultural runoffs sewage or other industrial pollutants, and algal blooms will result that could become toxic to fish, livestock and humans eating shellfish that feed on the phytoplankton and accumulate the toxins [12]. Moreover, the water must not be too warm, or too acid. All these conditions are deteriorating on account of global warming, bringing the prospect of a collapse in the marine biota, and turning the oceans into a massive carbon source that would further aggravate global warming (see other articles in this series).
We need urgent action to protect our oceans from pollution and over-exploitation. At the same time, we need to replace fossil fuels with the many renewable energy options that already exist (Which Energy?).
Article first published 21/07/06
Comments are now closed for this article